Attorney LLM
Attorney LLM helps attorneys do research of references, citators and the use of precedents to win cases. They can literally connect to databases such as PACER, USPTO, Unicourt or many state courts. Attorney LLM is designed to minimize the amount of hallucinations so attorneys can focus on winning strategies of their cases and not on spending hours looking for bibliography
LLM - RAG systems

LLM, RAG Systems
These systems are helping companies provide better customer service, provide support about procedures to workers in manufacturing, review specifications, help lawyers with references, review big stockpiles of data quickly and much more. By grounding Large Language Model (LLM) outputs in authoritative external data, RAG enhances explainability and traceability which are critical for regulatory compliance

Pharmaceuticals and Healthcare
Rag systems are used to evaluate the regulatory compliance of drug information and clinical trial protocols (PMC12917324). For example, systems like AskFDALabel extract adverse event terms and cardiotoxicity data directly from FDA-approved package inserts to ensure medical accuracy (PMC12917324). RAGs have to have traceability.
Automated Regulatory Labeling: RAG verifies that drug labels and promotional materials comply with the latest FDA guidance. The system flags "overstated benefits" or "missing warnings" by comparing the draft text directly against the approved clinical study report
CMC (Chemistry, Manufacturing and Controls) RAG helps maintain the "Golden Thread" of data from the lab to the factory floor, ensuring that he manufacturing process described in a regulatory filing matches the actual production logs.
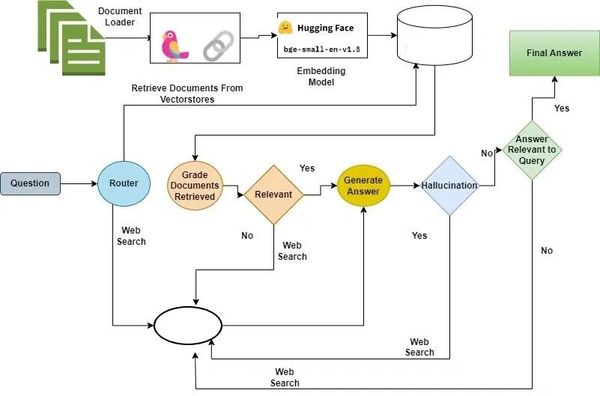
Legal Technology
RAG is applied to legal research, contract review, and predicting legal judgment. It allows for the rapid adaptation of recent legal precedents that were not included in the model's original training data. It can be designed to avoid hallucinations by adding extra logical loops which check for existence and topic validation of all given references.

Oil and Gas - Subsurface "Memory"
The RAG system ties the information contained in the historic legacy data with the real time sensor streams.
Subsurface Data Synthesis:
Geoscientists can use RAG systems to query unstructured drilling logs, seismic reports and lessons learned documents and get to know why a specific well failed back in the day so that it doesn't happen again.
Predictive Maintenance Guides:
When sensors flag a high pressure anomaly on a deep water rig, RAG instantly retrieves the specific technical manual and past maintenance tickets for that exact equipment serial number, providing engineers a step by step fix.
HSE (Health Safety and Environment) compliance:
Automating the cross referencing of new project plans against local environmental regulations and internal safety protocols to flag potential violations in seconds.

Oil and Gas - Subsurface "Memory"
The RAG system ties the information contained in the historic legacy data with the real time sensor streams.
Subsurface Data Synthesis:
Geoscientists can use RAG systems to query unstructured drilling logs, seismic reports and lessons learned documents and get to know why a specific well failed back in the day so that it doesn't happen again.
Predictive Maintenance Guides:
When sensors flag a high pressure anomaly on a deep water rig, RAG instantly retrieves the specific technical manual and past maintenance tickets for that exact equipment serial number, providing engineers a step by step fix.
HSE (Health Safety and Environment) compliance:
Automating the cross referencing of new project plans against local environmental regulations and internal safety protocols to flag potential violations in seconds.

Manufacturing - The Digital QMS
Manufacturing RAG focuses on operational efficiency and knowledge transfer to all workers
Standard Operating Procedures: technicians may have terminals to ask questions like how to recalibrate an instrument, and then the AI provides such procedure.
Supply Chain: RAG can monitor supplier and shipping delays and then suggest alternative vendors when parts are stuck in a port.
Quality Control: When a defect is found ,the RAG can search the Historical Defect Database and find reasons and solutions
This website uses cookies.
We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.